 For example, it cannot explain the differences in the properties of the two carbon allotropes – diamond and graphite. For example, argon and calcium have an atomic mass of approximately 40 amu.ĭalton’s theory could not explain the different allotropes of the same element. Previously, an atom was defined as the smallest part of an element that maintains the identity of that element. 4 ), is a fundamental concept that states that all elements are composed of atoms. However, it was found that even atoms of different elements can have the same mass. The modern atomic theory, proposed about 1803 by the English chemist John Dalton (Figure 1.5.4 1.5. For example, hydrogen, deuterium, and tritium are isotopes with different masses.ĭalton states that atoms of different elements can have different masses. However, it was later established that atoms of the same element can have different masses. However, we know an atom consists of electron, proton, and neutron.ĭalton states that atoms of a given element have precisely the same masses. Does not account for subatomic particlesĭalton stated that atoms are indivisible and cannot be further divided. While Dalton’s atomic theory laid the path toward the fundamental understanding of atoms and matter, it had a few demerits. However, both the sodium and chlorine atoms still exist. In the above example, sodium and chlorine combine to make salt. A chemical reaction is a rearrangement of atoms.ĭalton suggested that chemical reactions neither destroy nor create atoms. When they react, the Na and Cl atoms combine in a 1:1 ratio to form white crystals of sodium chloride (NaCl).Ĥ. The other is chlorine (Cl), which is a gas. One is sodium (Na), which is a highly reactive metal. For example, household common salt comprises two elements with varying physical and chemical properties. Compounds are formed from the combinations of two or more different types of atoms.ĭalton proposed that compounds comprise two or more atoms of different types. For example, an oxygen atom is different from a carbon atom.ģ. However, no two elements can have the same set of properties. 
Atoms of different elements differ in size, mass, and other properties. These concepts form the basis of chemistry. Atoms combine in whole-number ratios to form compounds. Atoms of the same element are the same atoms of different elements are different. It consists of three parts: All matter is composed of atoms. Atoms of a given element are identical in size, mass, and other properties. , first stated by John Dalton, an English scientist, in 1808. 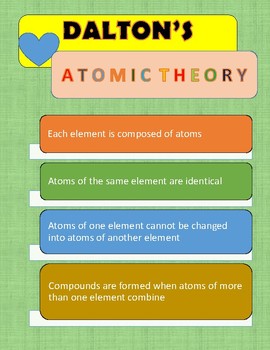
Atoms cannot be subdivided, created, or destroyed. The general tenets of this theory are: All matter is composed of extremely small particles called atoms. 
Elements may share similar properties like boiling and melting points and electronegativity. Dalton proposed his atomic theory in 1804. All atoms of a given element are identical in mass and properties.ĭalton proposed that every single atom of a specific element is the same as that of every other element. Dalton proposed that all matter is made of tiny indivisible atoms, which he imagined as “solid and movable particles”.Ģ. The laws of conservation of mass and definite proportions can be explained using the concept of atoms. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
